In-Vitro colorectal cancer screening tests market to accrue commendable receipts owing to a favorable regulatory structure, U.S. to lead the regional growth landscape
The growing number of technological advancements in cancer screening techniques have underlined the significance of in-vitro colorectal cancer screening tests market across the ever-evolving healthcare and medical devices industry. At the same time, the increasing demand for non-invasive cancer diagnosis methods has been a pivotal factor to have created a slew of business opportunities for companies operating in this vertical. With an overall valuation of about USD 785 million in the year 2016, in-vitro colorectal cancer screening tests market is projected to proliferate at an exceptional pace in the times to come.
China In-Vitro Colorectal Cancer Screening Tests Market, By Test Type, 2013 – 2024 (USD Million)
The latest trends affecting in-vitro colorectal cancer screening tests industry: How a slew of regulatory approvals have shaped the expanse of this business sphere
With an exclusive focus on the early detection and prevention of colorectal cancer, prominent in-vitro colorectal cancer screening tests industry contenders have, of late, focused on unveiling easy-to-use and patient-friendly screening assessments and test kits. Apparently, these tests have been proving to be beneficial for people of the age group of 50 and older who are at an average risk of developing colorectal cancer. Moreover, numerous innovative tests have been well-received by the healthcare regulatory authorities in the recent past which has had an optimistic influence on the overall growth potential of in-vitro colorectal cancer screening tests market. Enumerated below are a few noteworthy instances of regulatory approvals:
- In August 2014, the Wisconsin based molecular diagnostics company Exact Sciences Corporation declared that the Food and Drug Administration (FDA) of the U.S. has approved it’s noninvasive, stool DNA colorectal cancer screening test, Cologuard. As has been reported, the test has been formulated to detect biomarkers from DNA in cancer that is shed from the colon as part of the blood released in the stool and the overall digestive process. Furthermore, the test has been proven to find 69 percent of the most advanced precancerous polyps. Cologuard has also been recognized across the in-vitro colorectal cancer screening tests market as the first noninvasive screening assessment that analyzes both blood biomarkers and stool DNA.
- One of the major providers of sample and assay technologies for pharmaceutical research, molecular diagnostics, applied testing, Qiagen had announced in 2012 that the U.S. FDA has approved its genetic test. Reportedly, the therascreen KRAS RGQ PCR Kit would assist physicians to decide if cetuximab would be a conclusive treatment for colorectal cancer based on the lack of a gene mutation. It would be prudent to mention that cetuximab is conventionally known to target epidermal growth factor receptor on colorectal cancer cell surfaces and interrupts a signal preventing cell growth. Studies have shown that KRAS provides information about the KRAS mutation in patients whose colorectal cancer has metastasized – a development that is anticipated to bring about a paradigm shift across in-vitro colorectal cancer screening tests industry.
Get a Sample Copy of this Report:@ https://www.gminsights.com/request-sample/detail/451
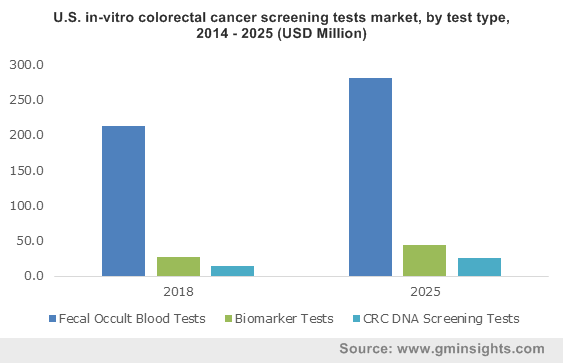
Speaking of the geographical growth outlook of this business sphere, the United States has persisted to be the forerunner across the North America in-vitro colorectal cancer screening tests market in 2016. Owing to unhealthy dietary habits, sedentary lifestyle, and rising levels of alcohol consumption, the prevalence of colorectal cancer among the residents of the nation has aggravated in the recent past. Moreover, due to the lack of patient compliance with screening tests recommended by healthcare professionals, colorectal cancer has emerged as one of the leading causes of cancer-related deaths in the U.S. According to reliable estimates, around 23 million Americans between the age of 50 and 75 avoid getting screened each year.
With several private and public undertakings unveiling highly effective awareness programs pertaining to the significance of screening tests coupled with technological advancements in diagnostic methodologies, the commercialization scale of in-vitro colorectal cancer screening tests industry appears highly promising. In fact, according to a research report put together by Global Market Insights, Inc., the total revenue share of this business space is forecast to surpass the billion-dollar frontier by 2024.
Author Name : Saif Ali Bepari
